Canal Ray Tube Experiment - Discovery Of Proton. #DiscoveryOfProton #Topic #NCERT #ForAllClasses
Hellow guys, Welcome to my website, and you are watching Canal Ray Tube Experiment - Discovery Of Proton. #DiscoveryOfProton #Topic #NCERT #ForAllClasses. and this vIdeo is uploaded by Chemistry Lerners [KNM] at 2021-04-11T03:49:51-07:00. We are pramote this video only for entertainment and educational perpose only. So, I hop you like our website.
Info About This Video
| Name |
Canal Ray Tube Experiment - Discovery Of Proton. #DiscoveryOfProton #Topic #NCERT #ForAllClasses |
| Video Uploader |
Video From Chemistry Lerners [KNM] |
| Upload Date |
This Video Uploaded At 11-04-2021 06:49:51 |
| Video Discription |
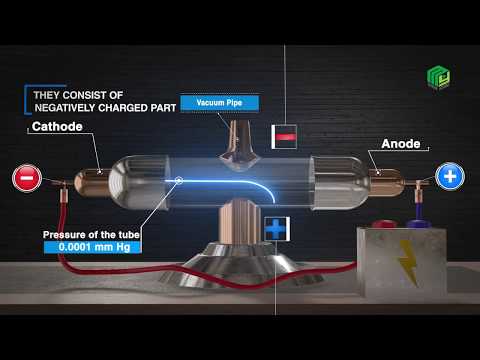
*Canal Ray Tube Experiment or Discovery Of Proton.
Discovery of proton was not done completely by canal ray tube experiment and This "Canal Ray Experiment" give the idea of positively charged particles. Canal ray tube experiment was first conducted by goldstein in 1886. "Eugen Goldstein" performed experiment and identify canal rays by modified cathode ray tube, the point of different difference was perforated cathode used. Canal ray tube is a tube made up of glass having two thin pieces of metal called electrodes they were sealed in it.one metal face act as cathode and other one act as anode. The electrical discharge is observed when only at very low pressures and at very high voltages. So the tube was evacuator, so that pressure of different gases could be adjusted. When sufficiently High voltage is applied across the electrodes current was passed through the both the electrodes creates an electric field in the tube and ionizes the gaseous atoms. Current start flowing through a stream of particles in the tube from the positive anode and negative cathode, These rays were called "canal rays" or "canal ray particles".
The rays move towards cathode and further passed to cathode strikes the glass wall, These rays further checked by coating the tube behind cathode with phosphorescent material "zinc sulphide". When these days after passing through cathode strike zinc sulphide coating red bright spots are developed on the coating.This bright spots are having positive charge , further called as "protons".
The characteristics of of positively charged particles are given as :-
(1) Unlike mass of positively charged particles depends upon on the nature of the gas present in the canal right tube, these are simply the positively charged gaseous ions.
(2) The charge to mass ratio of particles depends on the gas from which these originate.
(3) Some of the positively charged particles carry a multiple of the fundamental unit of electrical charge.
(4) The behaviour of these particles in the magnetic or electrical field is opposite to that observed per electrons or cathode rays. |
| Category |
Science & Technology |
| Tags |
Science & Technology Download MP4 | Science & Technology Download MP3 | Science & Technology Download MP4 360p | Science & Technology Download MP4 480p | Science & Technology Download MP4 720p | Science & Technology Download MP4 1080p |
More Videos